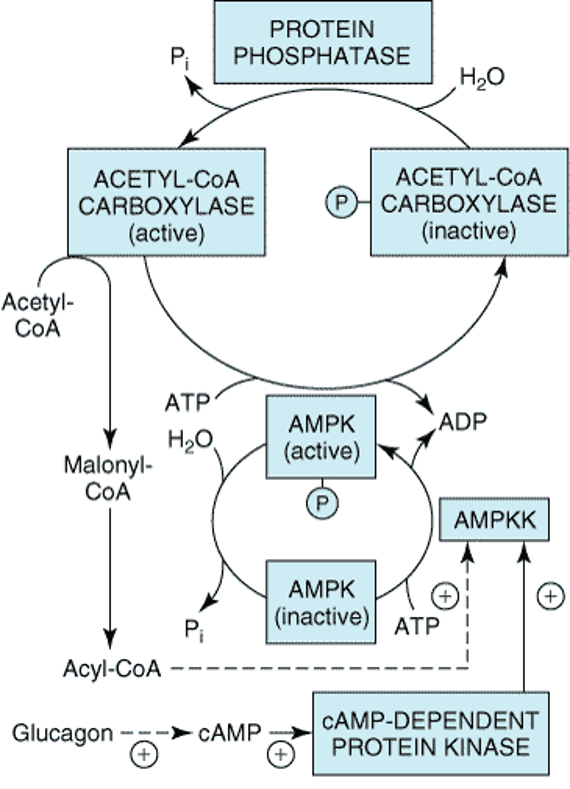
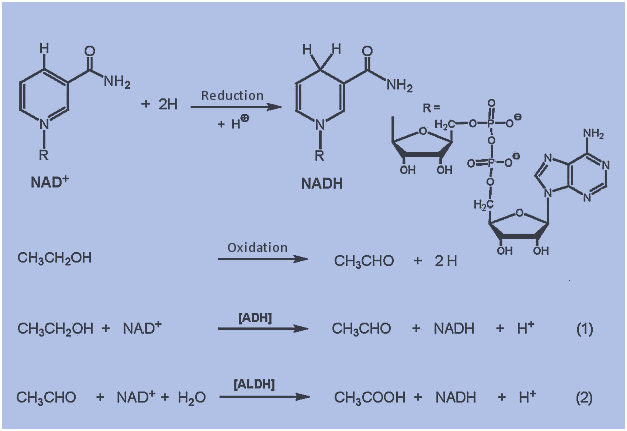
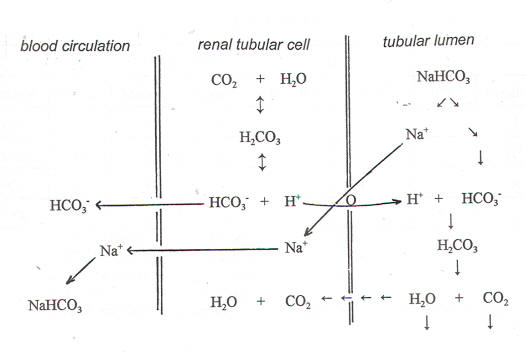
ATP- The energy currency within the cell
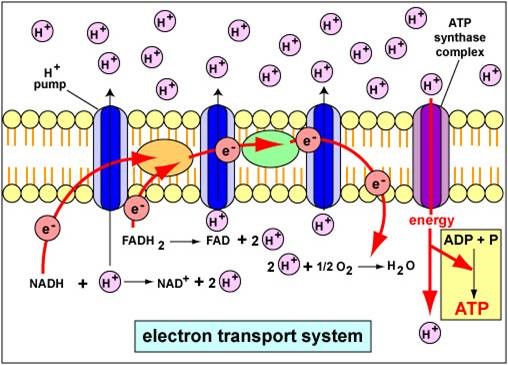
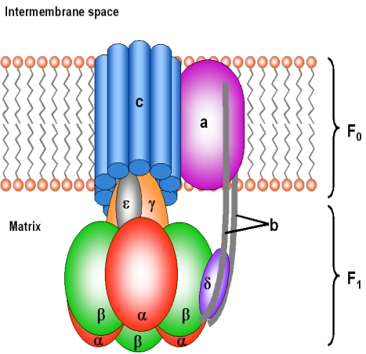
Q.- Adenosine triphosphate (ATP) is the energy currency within the cell. Which of the following best describes the type of compound ATP is? A. Phospholipid B. Amino acid C. Nucleotide D. Triacylglycerol E. Oligosaccharide. Details The correct answer is -C- Nucleotide. The nucleotide coenzyme adenosine triphosphate (ATP) is the most important form of chemical energy […]
ATP- The energy currency within the cell Read More »